

Welcome to the Scrambler Therapy® official scientific and clinical information site
The ST-NET project is based on an international automated network free for Scrambler Therapy® users. This network has been set up for various purposes, but its main one is to support clinical trials and pilot centers in reducing to a minimum operator-dependent variability results. By so doing, a quality system is created that, regardless of clinical trials chosen by researchers, guarantees reproducibility and consistent results not otherwise achievable, therefore reliable scientific clinical trials.
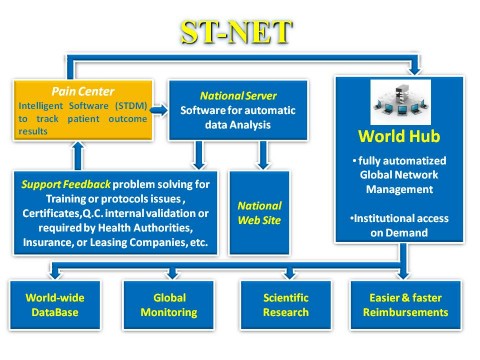 ST-NET is focused on a clinical “intelligent” folder based on
dedicated software designed exclusively for Scrambler Therapy.
Scrambler Therapy Data Manager (STDM) software manages in a simple
and effective way patients medical records in an easy-to-use
visual database. During compilation, STDM automatically informs
the operator of possible critical features and suggests the most
fitting treatment protocols for any patient and enables the use of
specific research tools such as the Brief Pain Inventory, Pain
Detect, DN4 and much more. This software can simultaneously (while
you are working on the record) open patient's data on other
traditional software such as Excel, Word or simply Notepad. It is
also possible to analyze treatment trends with graphs that do not
require previous configuration. The entire database can be
exported in Excel with filtering criteria based on data, pain
type, or other meaningful characteristics in clinical trials.
ST-NET is focused on a clinical “intelligent” folder based on
dedicated software designed exclusively for Scrambler Therapy.
Scrambler Therapy Data Manager (STDM) software manages in a simple
and effective way patients medical records in an easy-to-use
visual database. During compilation, STDM automatically informs
the operator of possible critical features and suggests the most
fitting treatment protocols for any patient and enables the use of
specific research tools such as the Brief Pain Inventory, Pain
Detect, DN4 and much more. This software can simultaneously (while
you are working on the record) open patient's data on other
traditional software such as Excel, Word or simply Notepad. It is
also possible to analyze treatment trends with graphs that do not
require previous configuration. The entire database can be
exported in Excel with filtering criteria based on data, pain
type, or other meaningful characteristics in clinical trials.
By clicking on “ Automatic analysis ” an algorithm analyzes the treatment data and highlights the main control parameters, indicating the percentage shift from the normal values. Meaningful shifts or serious anomalies are highlighted.
ST-NET is fully compliant with HIPAA and GDPR (General Data Protection Regulation) privacy standards. Implemented multi-level protection requirements are higher than those in most popular commercial software.
Contact Download Video Tutorial Delta Research & Development
Copyright © Delta Research & Development. All rights reserved